Learn About Flowflex COVID-19 At Home Antigen Test
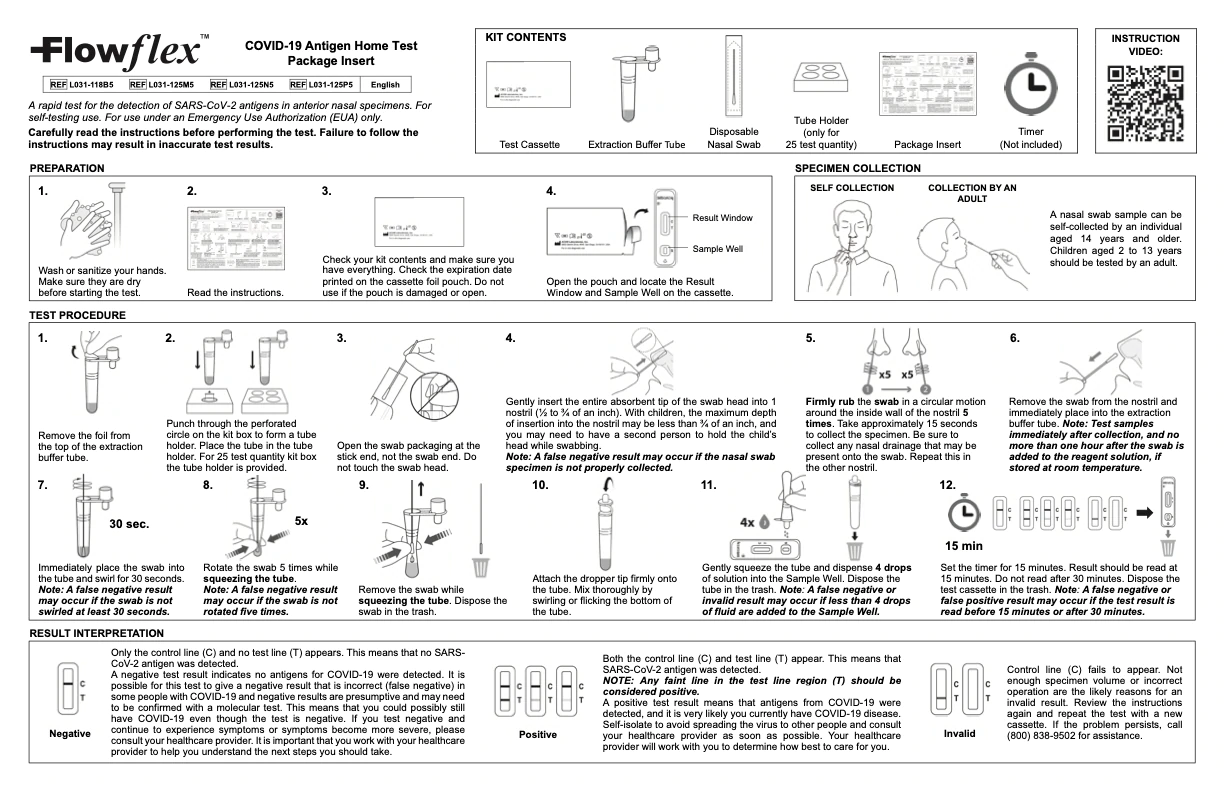
The FlowFlex COVID-19 antigen test is a rapid test used for the detection of SARS-CoV-2 antigens in anterior nasal specimens. Intended for self-testing use at home and available for use under an Emergency Use Authorization (EUA) only. Carefully read the instructions before performing this COVID-19 test on you or a loved one. Failure to follow the instructions may result in inaccurate test results.
ACON Laboratories, Inc. is the only legal manufacturer of the FDA EUA Flowflex COVID-19 Antigen Home Test.
This COVID-19 test product has not been FDA cleared or approved but has been authorized by FDA under an EUA.This product has been authorized only for the detection of proteins from SARS-CoV-2, not for any other viruses or pathogens.The emergency use of this product is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of IVDs for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug and Cosmetic Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated, or authorization is revoked sooner.
Flowflex COVID-19 At Home Antigen Test Overview:
- Easy-to-use nasal swab test
- Requires just 1 test
- Can be used to test children as young as 2 years old
- For use with and without COVID-19 symptoms
- Accurate results in 15 minutes
- No need to send off to a lab to obtain results
- Compact packaging for “On-The-Go” testing


















Reviews
There are no reviews yet.